Understanding the difference between polar and non-polar solvents is crucial for industries, including those using high-quality CDA-12A. These solvents are used in processes ranging from manufacturing to pharmaceuticals. This blog post aims to demystify these solvents. By the end, you’ll understand their roles and how they impact different industrial applications.
What Are Polar Solvents?
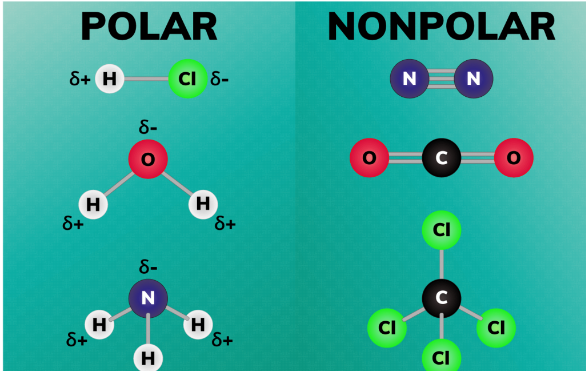
Polar solvents have molecules with a significant electronegativity difference between atoms. Water is the most common example. These solvents can dissolve ionic compounds and other polar substances. Their high dielectric constant makes them excellent for many chemical reactions.
Examples of Polar Solvents
Water is the most well-known polar solvent, celebrated for its ability to dissolve many substances due to its unique molecular structure. Other notable polar solvents include ethanol, methanol, and acetone, each possessing distinct properties that make them suitable for different applications.
Despite their differences, all these solvents share the crucial ability to dissolve polar substances, making them indispensable in various sectors such as pharmaceuticals, chemical manufacturing, and research. Their versatility and effectiveness in facilitating chemical reactions and processes highlight their importance in everyday life and specialized industries.
Industrial Applications of Polar Solvents
Polar solvents are used in drug formulation in the pharmaceutical industry. They help dissolve active ingredients efficiently. In chemical manufacturing, they facilitate reactions by dissolving reactants. The textile industry also benefits from using them in dyeing processes.
What Are Non-Polar Solvents?
Non-polar solvents have molecules with similar electronegativity values, resulting in an even electrical charge distribution. They are generally hydrophobic, meaning they don’t mix well with water but are excellent for dissolving non-polar substances.
Examples of Non-Polar Solvents
Common non-polar solvents include hexane, benzene, and toluene. These solvents are usually hydrocarbons. They are not miscible with water but are excellent at dissolving oils and fats, making them essential in various industrial processes.
Industrial Applications of Non-Polar Solvents
Non-polar solvents are widely used in the petrochemical industry. They help extract and refine oils. The paint industry uses them as carriers for pigments and resins. They also play a role in the production of adhesives and sealants.
Comparing Solubility
Polar solvents can dissolve ionic and other polar substances, while non-polar solvents dissolve non-polar substances like oils and fats. This difference is due to the “like dissolves like” principle. Understanding this helps in selecting the right solvent for specific applications.
Impact on Chemical Reactions
Polar solvents often act as catalysts in chemical reactions. They stabilize ions and polar molecules, facilitating faster reactions. Non-polar solvents, on the other hand, are inert. They do not participate in reactions but provide a medium for non-polar reactants.
Environmental Considerations
Both types of solvents have environmental impacts. Polar solvents like methanol are biodegradable but can be toxic. Non-polar solvents like benzene are less biodegradable and are considered hazardous. Proper disposal and handling are crucial.
Safety Measures
Handling polar and non-polar solvents requires safety precautions. Polar solvents are often flammable and toxic. Non-polar solvents are also flammable and can be carcinogenic. Always use protective gear and follow safety guidelines.
Future Trends in Solvent Use
The trend is shifting towards greener alternatives as industries recognize the importance of sustainability. Companies are actively exploring bio-based solvents from renewable resources to reduce environmental impact while maintaining process efficiency. These solvents minimize harmful emissions and support a circular economy using replenishable materials.
Additionally, advancements in producing and applying these bio-based solvents ease the transition, enabling businesses to adopt eco-friendly solutions without sacrificing performance or productivity. Consequently, the movement towards greener practices is gaining momentum, fostering a healthier planet for future generations.
Conclusion
Polar and non-polar solvents, including high-quality CDA-12A, play vital roles in various industries. Understanding their properties and applications can help in making informed decisions. These solvents are indispensable in pharmaceuticals, petrochemicals, and manufacturing. For more information, consider consulting experts in the field.